

This therapist directory is offered in partnership with BetterHelp. If you sign up for therapy after clicking through from this site, HelpGuide will earn a commission. This helps us continue our nonprofit mission and continue to be there as a free mental health resource for everyone.
Need to talk to someone now? Find a crisis helpline
If you're a BetterHelp therapist with questions about your directory listing, please contact therapists@betterhelp.com
There are many different treatments available for mental health issues, including therapy, medication, peer support, and self-help strategies. By better understanding your options, you can make the choices that are right for you.
View FAQs

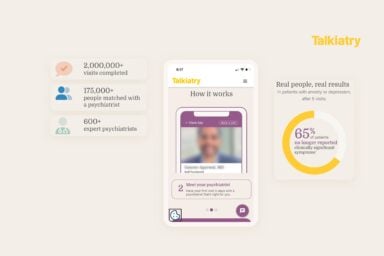
The HelpGuide team personally tested out Talkiatry and surveyed users to understand their experience with the platform. Here’s what we discovered about the online psychiatry platform.
Infidelity can devastate a marriage, but healing is possible. Learn how therapy can be beneficial following an extramarital affair.

Certain over-the-counter anxiety medications might help calm your mind. Explore the benefits, limitations, and risks of natural remedies and supplements.
We surveyed real couples who used Regain, and a HelpGuide staff member personally tested its services with a partner. Here’s what we learned about the couples therapy platform.
We surveyed real Sesame Care users and a HelpGuide staff member personally tested its mental health services. Here’s what we learned about the platform’s usability, provider quality, user satisfaction, and more.

Clonidine is a medication used to treat high blood pressure. But it might also be useful for managing anxiety. Learn more about how this medication works and its side effects.
Prozac (fluoxetine) is an FDA-approved medication for anxiety-related conditions. Learn more about how this medication works and its side effects.
Lexapro (escitalopram) is an FDA-approved medication for long-term anxiety treatment. Learn more about how this medication works and its side effects.